Very low-density lipoprotein receptor (VLDLR)-C-tag purification from HEK293E cells
Patricia Yuste-Checa, F Ulrich Hartl, Andreas Bracher
Abstract
This protocol details how to purify recombinant Very low-density lipoprotein receptor (VLDLR)-C-tag protein from HEK293E cells.
Attachments
Steps
VLDLR-C-tag expression
Express VLDLR-C-tag in HEK293E cells cultured in FreeStyle 293 Expression Medium for 96h 0m 0s
Centrifuge culture and keep conditioned medium.
Dialyze 300mL conditioned medium against 10L Binding buffer.
CaptureSelect C-tag affinity chromatography
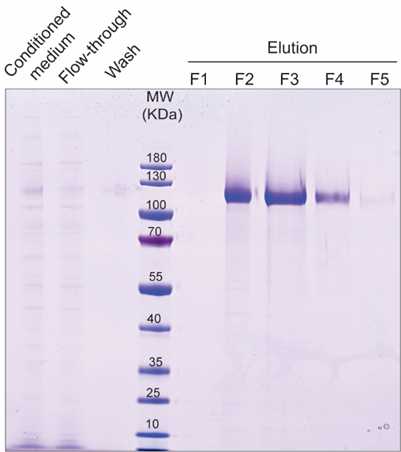
Load dialyzed conditioned medium onto a CaptureSelect C-tag affinity column previously equilibrated with binding buffer (column volume, CV: 4 mL slurry for 300 mL dialyzed media) by gravity flow at 4°C.
Wash the column with 5 CV of Binding buffer.
Elute VLDLR-C-tag protein with 6x 1 mL of Elution buffer. Collect fractions of 1mL.
Pool fractions containing VLDLR-C-tag protein.
Exchange protein buffer to Binding buffer with a NAP-25 desalting column previously equilibrated with Binding buffer.
Collect the fractions containing protein, concentrate by ultrafiltration to >1 mg mL-1, aliquot and flash-freeze purified VLDLR-C-tag in liquid nitrogen for storage at -70°C.