Labelling kelps with 13C and 15N for isotope tracing or enrichment experiments
Anton Kuech, Ursula Witte, Inka Bartsch
Macroalgae
Kelp
Seaweed
Cultivation
Isotopic enrichment
Isotope tracing experiments
Enrichment experiments
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
Isotope tracing experiments can be used to trace organic material flow through the ecosystem by artificially adding labelled biomass into a system. The advantage of this process is the direct control of carbon and nitrogen addition to the system for measuring uptake rates by consumers which can substantially reduce the uncertainties associated with food web models. This protocol details the steps involved in successfully culturing and isotopically enriching (13C & 15N) juvenile sporophytes of two common North Atlantic kelp species (Laminariales): Saccharina latissima and Laminaria digitata . A first-order successful isotopic enrichment study of S. latissima , as well as the first inclusion of 15N enrichment for L. digitata , is detailed. This protocol provides a comprehensive description of the stable isotope enrichment process in two kelp species, potentially serving as a foundation for its application in other macroalgal taxa.
Before start
The generation of juvenile sporophyte material for isotopic labelling can be achieved through various methods, such as sampling from the field or growing sporophytes from spores or vegetative stock gametophytes. For details of our experiment see PLOS ONE article. Juvenile kelp sporophytes for labelling purposes have to be cultured in a way, which ensures good growth to produce actively growing healthy material. The following steps are tailored to the two species cultivated for this experiment and may require adaptation based on the available equipment and sporophyte physiology.
Attachments
Steps
Culture set-up (pre-labelling)
Sterile culture bottles (glass or plexiglass) of sufficient size are used for cultivation. In our case, sporophytes of Laminaria digitata and Saccharina latissima were cultured in 5L DURAN glass bottles with the nutrient addition of 100mL (half concentration) Provasoli Enriched Seawater (PES) in 10L fresh seawater stored in temperature-controlled laboratories at 10°C. Medium was changed weekly and continuously bubbled with dry air via tubes. Irradiance should be set to around 40-50 µmol photons m-2s-1in long-day conditions (16 h light : 8 h dark; 16:8 LD). For cultivation of sporophytes, clean and pasteurised seawater is needed, either from natural fully marine sources or artificial seawater at approximately 30-33 PSU. Here, seawater was filtered through a 5" Polypropylene Yarn Water Filter and was pasteurised for 4h 0m 0s at 99°C with a combi steamer (PALUX, Germany).
Provasoli Enriched Seawater (PES) medium solution
Equipment
| Value | Label |
|---|---|
| DURAN™ Original Laboratory Bottle, Clear, with DIN 168-1 Thread, Graduated | NAME |
| DURAN glass bottle | TYPE |
| DWK Life Sciences | BRAND |
| Z232122-1EA | SKU |
| https://www.fishersci.com/us/en/home.html | LINK |
| 5000 mL (narrow neck) | SPECIFICATIONS |
Equipment
| Value | Label |
|---|---|
| 5" Polypropylene Yarn Water Filter | NAME |
| Water Filter | TYPE |
| Vyair | BRAND |
| n.a. | SKU |
Equipment
| Value | Label |
|---|---|
| PALUX Touch 'n' Steam 611QL | NAME |
| Combi Steamer | TYPE |
| PALUX | BRAND |
| E611 QBRN 000000 | SKU |
Determining growth rates
Macroalgal growth rates should be determined in simulated labelling conditions prior to labelling start to ensure the material is growing well. Otherwise, successful incorporation of labelled compounds is not guaranteed. Wet weight (mg) and surface area (cm2) are the main variables that should be measured at regular intervals (e.g. every 4 days), but blade length can also be used as a variable. In addition, pH measurements can be taken to monitor the acidity of the media.
Labelling medium
The simulated medium for labelling consists of von Stosch (VS) medium (in contrast to PES, which is used during early cultivation – see Note below) modified to exclude phosphate and nitrate. Phosphate (0.01546millimolar (mM) 0.54908millimolar (mM) 2.32545millimolar (mM)
| A | B | C | D |
|---|---|---|---|
| Chemical compound | CAS number | Concentration (μmol/L) [as in full PES] | Concentration in millimolar (mM) |
| Sodium bicarbonate | 144-55-8 | 2325.45 μmol/L | 2.32545 |
| Sodium nitrate | 7631-99-4 | 549.08 μmol/L | 2.32545 |
| Sodium glycerophosphate – hydrate | 55073-41-1 | 15.46 μmol/L | 0.01546 |
Chemical concentrations added individually to von Stosch (VS) medium used for cultures (pre-labelling) with CAS Registry Number of chemical substances.
Sporophyte preparation for labelling
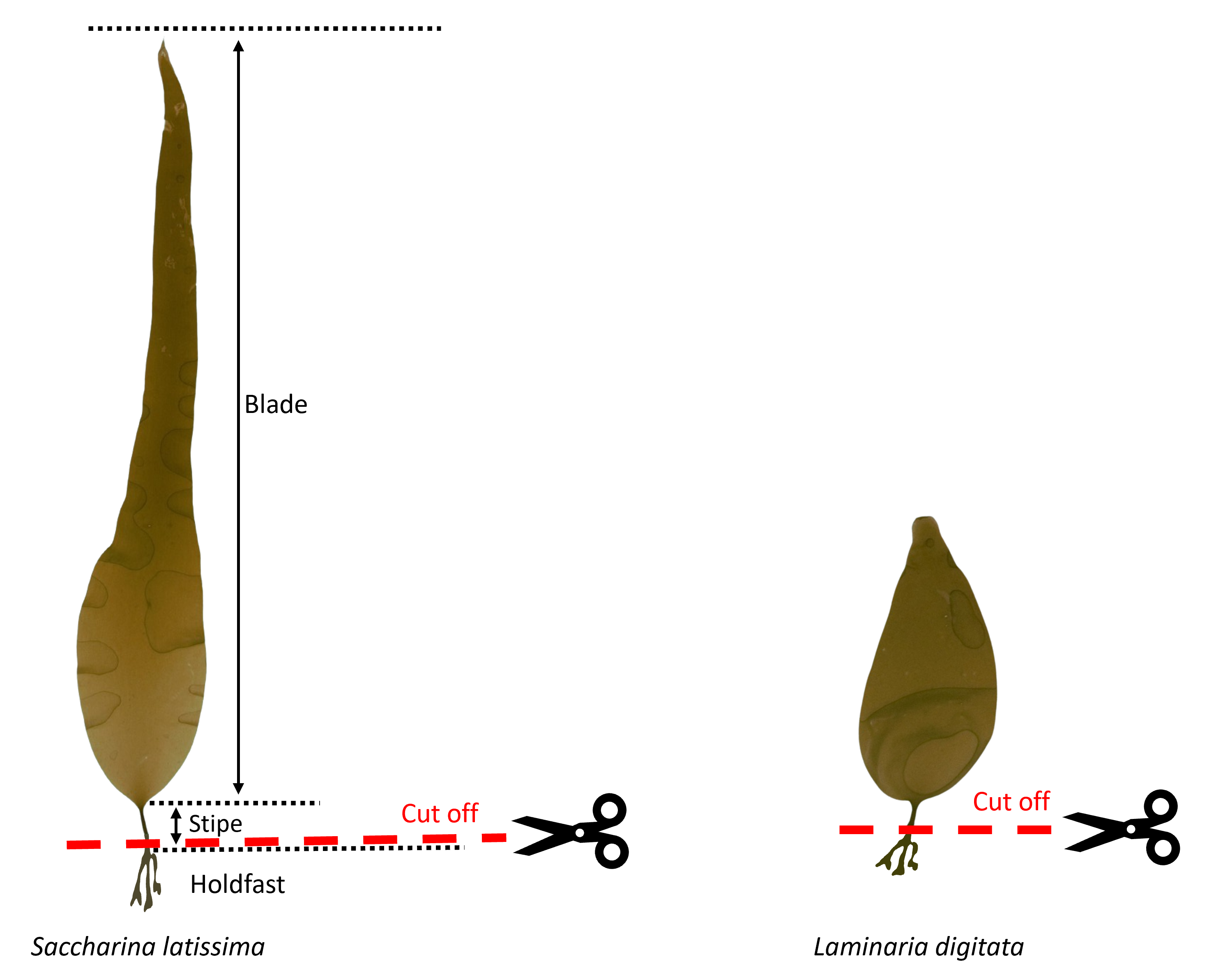
Separate sporophytes of the two species by cutting them at the stipe just above the holdfast using dissecting scissors. This enables single unidirectional growth as compared to an unequal distribution of growth between blade and holdfast. Select sporophytes of a good growth stage (e.g. widening of blade for S. latissima ) and avoid individuals with white spots or other noticeable damaged areas. The blade should have an even brown colour.
Equipment
| Value | Label |
|---|---|
| Fisherbrand™ Dissecting Scissors | NAME |
| Scissors | TYPE |
| Fisherbrand | BRAND |
| 15277168 | SKU |
Example incubation of sporophytes for determining growth rates
Either two sporophytes of Saccharina latissima or four sporophytes of Laminaria digitata were placed into a single 2L Kautex bottle (ensure sufficient replication). Wet weight (mg), surface area (cm2) and pH were measured in 4-day intervals for a 14-day period.
Equipment
| Value | Label |
|---|---|
| Wide Necked square container, PETG, 2000mL w/ screw closure, crystal clear | NAME |
| Wide neck container | TYPE |
| Kautex | BRAND |
| 225274374 | SKU |
Labelling of kelps
Pre-labelling start conditions
A few days prior to labelling start, full PES (200mL PES in 10L pasteurized seawater) should be added and irradiance should be set to 50 µmol photons m-2s-1to induce optimum growth rates (16 h light : 8 h dark per 24-hour period).
Labelling conditions
The nutrient concentrations used in von Stosch (VS) were adjusted to represent the same concentration that the macroalgae would receive in the full PES mixture (200mL PES in 10L pasteurized seawater). This concentration is considered the ideal medium for growth and is also in accordance with the concentrations used by Braeckman et al. (2019) and Rossi et al. (2013). Exact concentrations of chemicals added separately were 0.01546millimolar (mM) 0.55457millimolar (mM) 2.35294millimolar (mM)
| A | B | C | D |
|---|---|---|---|
| Chemical compound | CAS number | Concentration (μmol/L) [as in full PES] | Concentration in millimolar (mM) |
| Sodium glycerophosphate – hydrate | 55073-41-1 | 15.46 μmol/L | 0.01546 |
| Sodium nitrate-15N | 31432-45-8 | 554.57 μmol/L | 0.55457 |
| Sodium bicarbonate-13C | 87081-58-1 | 2352.94 μmol/L | 2.35294 |
Concentrations of main chemicals used for labelling with CAS Registry Number of chemical substances.
Von Stosch (VS) enriched seawater medium solution
Separate sporophytes of the two species by cutting them at the stipe just above the holdfast using nail scissors as detailed in step #4.
Incubation of sporophytes for labelling
Selected kelp sporophytes are stored in Kautex bottles, filled with the pasteurized seawater and labelling solution to the top, tightly closed and placed on rotating or tilting shakers for 9 days. Kautex bottles lie on their sides to allow full illumination. Make sure that there are no air bubbles in the bottle. We used Orbitron rotating shakers at 90rpm. Irradiance set to 50 µmol photons m-2s-1in a 16 : 8 LD cycle.

Equipment
| Value | Label |
|---|---|
| Wide Necked square container, PETG, 2000mL w/ screw closure, crystal clear | NAME |
| Wide neck container | TYPE |
| Kautex | BRAND |
| 225274374 | SKU |
Equipment
| Value | Label |
|---|---|
| Orbitron | NAME |
| Shaker | TYPE |
| INFORS HT | BRAND |
| n.a. | SKU |
Harvesting biomass
Remove sporophytes from a single Kautex bottle.
Take picture and wet weight of sporophytes using a macro photography camera or similar.
Separation of growing from non-growing blade parts
After the incubation period, newly formed blade parts of sporophytes normally become visible by a widening at the basis (see image below). We expected that the basal blade part that was formed during incubation would have a higher labelling result than the distal blade part. This assumption was supported by our results (see PLOS ONE article for details). Thus, if you cut sporophytes at the intersection between the estimated new growth (highly labelled) and prior biomass (slightly labelled), you separate material with a differential labelling result.

Dip sporophytes briefly in deionized (DI) water to remove residual saltwater and dry material with clean tissue paper.
Shock freezing of samples
Sporophytes wrapped in punctured aluminium foil (highly/slightly labelled separate) and either (a) dipped in -80°C prior to freeze-drying.
Freeze-drying of samples for 48 hours.
Equipment
| Value | Label |
|---|---|
| RVC Alpha 3-4 LSCbasic | NAME |
| Laboratory freeze-dryer | TYPE |
| CHRIST | BRAND |
| n.a. | SKU |
Storage of freeze-dried biomass in zip lock bags in freezer at -20°C (or -80°C). Material can also be stored in a desiccator filled with silica gel for shorter periods.
Determining labelling uptake
Freeze-dried material should ideally be made into powder, best with a ball mill. Remove sporophytes from foil and transfer into the metal tubes from the ball mill. Ball mill set to 0h 3m 0s at 25 Hz. Afterwards, store the powder in combusted glass vials (previously combusted at 500°C overnight).
Equipment
| Value | Label |
|---|---|
| Mixer Mill MM 400 | NAME |
| Retsch | BRAND |
| MM400 | SKU |
Equipment
| Value | Label |
|---|---|
| Fisherbrand™ Snap Cap Vial, Clear Glass | NAME |
| Glass vial | TYPE |
| Fisherbrand | BRAND |
| 10749644 | SKU |
| 14 mL volume | SPECIFICATIONS |
Random sub-samples of the powder, such as 4 replicates per species and distal/basal blade part, are weighed into aluminium cups (approximately 1mg per sample), encapsulated and added onto a 96 well plate (can also be stored in a desiccator filled with silica gel for shorter periods).
Equipment
| Value | Label |
|---|---|
| Aluminium capsules/pans for solids | NAME |
| Aluminium capsules | TYPE |
| Elemental Microanalysis | BRAND |
| D3089 | SKU |
| Aluminium Capsules Pressed 8.75 x 3.5mm pack of 100 | SPECIFICATIONS |
Equipment
| Value | Label |
|---|---|
| Corning™ Clear Polystyrene 96-Well Microplates | NAME |
| 96-well plate | TYPE |
| Corning | BRAND |
| 10377601 | SKU |
Analysis of samples for 13C and 15N isotopes. For details of the results for this experiment see PLOS ONE article.

