PURIFICATION OF PROTEINS FROM PFA FIXED SAMPLES BAK_WITH BIOTIN PULLDOWN FOR LC-MS/MS_2023
Bryan Killinger
Abstract
This protocol details the purification of proteins from PFA fixed samples and extraction of proteins from formalin-fixed tissues. Also included, biotin pulldown prior to LC-MS/MS. Samples generated for this protocol have been used for mass spectrometry, immunoblotting, and pulldowns.
Attachments
Steps
Procedure Day 1 (Extract proteins and test concentration)
Wash sections in DM 3 X 10 min.
Wash sections in DM for 0h 10m 0s (1/3)
Wash sections in DM for 0h 10m 0s (2/3)
Wash sections in DM for 0h 10m 0s (3/3)
Place sections in 1.5mL Eppendorf tube.
Add 0.5mL of reversal buffer.
Briefly sonicate on low power to disperse tissue.
Add 5µL of 100millimolar (mM) PMSF. Mix well. Quick spin.
Heat on block for 98°C for 0h 30m 0s with cap locks.
Remove from heat block carefully (caps will pop if not careful).
After 0h 5m 0s of cooling, vortex well.
Centrifuge at 22000x g,0h 0m 0s for 0h 30m 0s Room temperature.
Collect S1 (extracted proteins).
Western blot (Optional)
Perform methanol/chloroform cleanup on 100µL of S1.
Resuspend resulting pellet in 100µL 5% SDS.
Perform BCA assay on 100µL. Each BCA test takes 20µL. The remaining volume can be used for western blot. Alternatively, more protein can be prepared from S1 if needed.
Calculate volume required for 20µg protein.
20ug / concentration of S1 ug/ul = ul of sample required
Procedure Day 2 (Capture biotinylated proteins and wash)
Add S1 to 10mL TBST in 15mL conical tube. Mix well.
Add 40µL of prepared magnetic streptavidin beads to each tube.
Nutation for 1h 0m 0s Room temperature.
Place tube on magnetic stand for 0h 1m 0s (until all beads have been drawn out of solution).
Carefully remove supernatant no to disturb beads. Add 10mL wash buffer.
Nutation for 0h 30m 0s.
Place tube on magnetic stand for 0h 1m 0s (until all beads have been drawn out of solution).
Add 10mL wash buffer.
Nutation for 0h 30m 0s.
Place tube on magnetic stand for 0h 1m 0s (until all beads have been drawn out of solution).
Add 10mL wash buffer
Nutation 0h 1m 0s at 4°C.
Day 3 (Elute captured proteins)
Place tube on magnetic stand for 0h 1m 0s (until all beads have been drawn out of solution).
Carefully remove supernatant no to disturb beads.
Add 1mL of wash buffer.
Mix samples until beads are suspended in wash buffer.
Using a pipette with tip cut off, transfer suspended beads to a 1.5mL protein low bind tube.
Place tube on magnetic stand for 0h 1m 0s (until all beads have been drawn out of solution).
Remove buffer and surface wash with milliq 2X.
Quick spin and remove liquid from tube (should only have beads left).
Add 80µL 1X SDS-page sample buffer containing reducing agent.
Mix well and quick spin.
Place on heat block set to 98°C for 0h 10m 0s.
Mix well and quick spin.
Place on magnet.
Transfer eluent to two separate lo-bind tube and discard used beads. 35µL in 1 tube (used for premass spec QC), the remaining 45µL in another (used for mass spec).
Immediately place in -80°C for long-term storage.
Day 4 (Prepare proteins for Mass Spec)
Prepare Bis-Tris 4-12% wedge gel 15 well.
Make 800mL 1X MOPS running buffer.
Load 5µL of MW standard in one side of gel.
For MS load exactly 40µL of the samples into wells. Careful not to spill into opposing wells. Only load samples into every other well to prevent cross contamination.
Ensure all wells have a buffer. For empty wells fill with 40µL 1X loading buffer.
Run the gel at 150V for a few minutes. Watch carefully until the sample has completely entered gel.
Stop running, remove gel with clean gloves, and clean equipment.
Fix gel in 100mL fixation solution (refer materials section) for 1h 0m 0s Room temperature.
Wash gel in several changes of milliq, until gel has swollen to original size.
Place in 100mL colloidal Coomassie blue stain solution. Cover loosely, and heat in microwave for few minutes until solution just starts to boil.
Incubate gel in heated colloidal Coomassie blue stain solution for 0h 8m 0s.
Remove gel from stain solution and wash several times with milliq water. Clear background is achieved with ~2h 0m 0s of washing.
Using a clean razor excise the entire sample from the gel.
Place each sample into a clean 1.5mL tube. Add 500µL of milliq water.
Samples can be stored at 4°C until submitted to mass spec core.
Optional: Pre mass spec QC
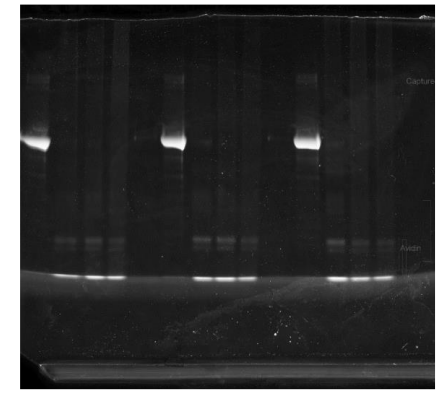
Estimating capture concentration - Purpose: To provide mass spec core with a rough estimate of the amount of protein in the capture sample.
Prepare gel as described above (Day 4, Steps 43-45)
Load 20µL of QC aliquot into wells.
Load 2µg BSA into 1 well.
Run the gel at 150V for approximately 0h 45m 0s or until the die has reached end of gel.
Stain gel with Coomassie as described above (Day 4, Steps 50-54)
Image on Odyssey using NIR.
Dot blot to estimate target enrichment
Cut PVDF membrane into ~1X3 inch piece.
Activate PVDF in 100% methanol for 0h 0m 30s
Equilibrate activated PVDF in diH20.
Just prior to blotting samples, place hydrated PVDF on wypall and quickly pat dry.
Place PVDF on new dry wypall.
Spot 1µL of each sample onto PVDF.
Allow PVDF to dry completely. (~1h or 0h 0m 30s)
Reactivate spotted PVDF as described above and equilibrate in dH20.
Blot can now be probed with antibodies using standard protocols.